A drug alert has been issued this week warning people with asthma to check their inhalers due to defective batches.
Specific batches of Ventolin Accuhaler inhalers, used for the emergency relief of asthmatic symptoms, are being recalled in the UK over a manufacturing issue.
A fault was detected in the inhalers that results in a small number of the devices not delivering the full number of doses.
Additionally, one lot of Seretide Accuhaler, used for the maintenance of preventative treatment of asthma, is being recalled from hospitals, pharmacies, dispensing practices, retailers and wholesalers in the UK.
The recall of Seretide Accuhaler is pharmacy level because this is used for maintenance treatment as opposed to a reliever treatment.
Thousands of products are said to be affected by the fault, which were distributed to the UK market.
The Medicines and Healthcare products Regulatory Agency (MHRA) issued the patient level drug alert recalling two specific affected lots manufactured by Glaxo Wellcome UK Limited.
“Patients who have used the affected batches of Ventolin may find that their symptoms are not relieved as normal by their Ventolin Accuhaler. If this happens, they should seek medical advice. Affected Accuhalers should be returned to their pharmacist for a replacement,” the alert said.
Only a small proportion of the units are defective. Other asthma inhalers, including the more commonly used Ventolin Evohaler, are not affected.
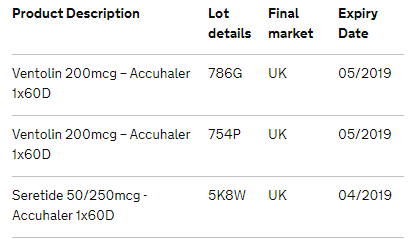
The affected product details are below:



Bernadette Sinclair Jenkins, MHRA’s Regulatory Assessment Unit Manager of the Inspections, Enforcement and Standards unit said: “It is important people check whether they have an affected inhaler. We want patients and their families to be confident treatment will be safe and effective when required.”
“People with a Ventolin Accuhaler from the affected lots should take them to their pharmacy or their dispensing practice and speak to a pharmacist who will provide a replacement.
“We strongly encourage anyone to report any issues with their inhalers or other medicines or medical devices to MHRA via our Yellow Card Scheme.
“Information is also available to patients and healthcare professionals by contacting GSK’s Customer Support Team via customercontactuk@gsk.com or calling on 0800 221 441 (option 4).”








